Best Coatings for Medical Devices
Posted by Sean Horn
Friday, November 6, 2020 8:00
@ 8:00 AM
Parylene is a polymeric material that is commonly applied as a conformal coating layer in electronic applications. Parylene conformal coatings are used to provide environmental and/or dielectric isolation. They offer pinhole-free layers with low water permeability, high flexibility and high mechanical strength (see table). While parylene coatings are most frequently applied onto the electronic circuitry and sensors they also have impeccable properties that are beneficial for use in medical substrates.
Use of Parylene as a packaging material for bio medical applications
Medical substrates are exposed to the bio chemical environment of the living organism (human, animals) and are prone to chemical attacks by various body fluids upon prolonged contact with human tissue at 37 °C and between pH 5.4–7.4 [1]. Therefore, corrosive biochemical environment has to be kept isolated. Reliable, hermetic packaging that does not trigger immune response is required to isolate electronic parts and systems that are found in the medical application.
Parylenes have been widely investigated for use in medical applications. Materials used in medical systems packaging interface with living tissue and biochemical liquids. Parylene C and N complies with the FDA regulations for biomedical use (ISO-10993 Biological Evaluations for Medical Applications microfluidic devices, medical implants (stents, needles, etc.) therefore they can used in bio-medical devices such as implants and skin patches that contact with the skin for data collection (eg. Pressure sensors for heartbeat, thermal sensors for body temperature, chemical sensors for sweat analysis, acoustic sensors in cochlear implants, etc). Medical applications ranging from neural prosthetic applications, catheters, stents to skin patches makes use of parylene conformal coatings [2]–[5].
Prior Studies on the bio-compatibility
Bio corrosion prevention properties of Parylene in biochemical environments have been investigated by the scientific community. Coatings for improving corrosion resistance of stainless steel 316L implant materials investigated the effect of bodily fluids on the 2 um thick Parylene N layer using Hanks solution [1]. It is an artificial salts mixture including blood serum, tissue extracts (see Image). In the absence of H2O2 (immune response) Parylene layer behaves as an ideal candidate for an implant conformal coating and in the study a deterioration mechanism was proposed for the cracked film. They reported that the deterioration starts from the metal/polymer interface and progress towards the outer surface resulting in cracks as seen in the image.
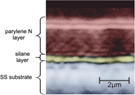

Cross section image of polymer coating on stainless steel (1st). SEM images of the polymer coated stainless steel before exposure (2nd ), after 7 days of exposure to Hanks solution (3rd ) and Hanks solution with H2O2 (4th ). (Image Ref: [1]).
In another study, Parylene C was studied using Hanks solutions and H2O2 to see its
compatibility with the bio chemical environment [6]. Parylene C coatings between 2–20 μm on stainless steel 316L implants were investigated it was concluded that an 8 proved to have excellent wear resistant properties limiting the wear debris formation efficiently.
Finally, a comparative study on Parylene N and C revealed that Parylene C coatings, the critical load for initial cracks is 3–5 times higher and the total metal ions release is reduced 3 times more efficiently compared to Parylene N [7].
A five months study on Parylene C as a packaging material on test chips to protect CMOS circuitry kept at 55°C in isotonic saline soak tests has shown that the conformal coating has not been affected by the soak test [8]. In, another study making use of 25 μm thick
Parylene C as a packaging material for neuro sensor implants (in vivo and in vitro) a preliminary assessment of the stability for in vivo implantation was shown. Parylene C stability as reported [9].
In conclusion, Parylene C is an ideal packaging material or a flexible substrate for bio medical implants. It has a well-known and established deposition process which also offers compatibility with standard microfabrication processes for use with MEMS based implants and skin patches. Particularly, its ISO class VI biocompatibility makes it a great candidate. Another advantage of parylene C conformal coating as a biomedical substrate package is its transparency and flexibility which offers ease of surgical manipulation.
Table: Parylene Types and Properties *Materials data: MatWeb
| Property | Unit | Parylene N | Parylene C | Parylene F |
| Molecular structure | ||||
| Chemical Formula | C16H16 | C16H14Cl2 | C16H8F8 | |
| Optical | Transparent | Transparent | Transparent | |
| Biocompatibility | Yes | Yes | Yes | |
| Electrical properties | ||||
| Dielectric constant (@ 1 MHz) | 2.66 | 2.95 | 2.17 | |
| THERMAL PROPERTIES | ||||
| Melting point | °C | 420 | 290 | ≤ 500 |
| Durable Heat Resistance | °C | 80 | 100 | 350 |
| Thermal conductivity | Cal/sec | 3 | 2 | |
| MECHANICAL/PHYSICAL PROPERTIES | ||||
| Water Absorption | 0.01%/24 hour | 0.06%/24 hour | 0.01%/24 hour | |
| Oxygen Transmission
(@Temperature 25.0 °C) |
cc-mm/m²-24hr-atm | 15.4
|
2.80
|
34.7 |
| Advantages | ü Constant dielectric coefficient at all frequencies
ü High dielectric strength ü Less wear (low friction coefficient.) |
ü Low gas permeability
ü High Chemical Resistance |
ü Sub-micron coverage [10]
ü High thermal resistance ü UV-resistive ü High-density |
|
If you’re looking for medical device coating services, contact Diamond-MT today. Get started by calling us at 814-535-3505 or completing a quote request.
References:
[1] M. Cieślik, K. Engvall, J. Pan, and A. Kotarba, “Silane–parylene coating for improving corrosion resistance of stainless steel 316L implant material,” Corros. Sci., vol. 53, no. 1, pp. 296–301, Jan. 2011, doi: 10.1016/j.corsci.2010.09.034.
[2] L. Wei, A. Lakhtakia, A. P. Roopnariane, and T. M. Ritty, “Human fibroblast attachment on fibrous parylene-C thin-film substrates,” Mater. Sci. Eng. C, vol. 30, no. 8, pp. 1252–1259, Oct. 2010, doi: 10.1016/j.msec.2010.07.003.
[3] P.-J. Chen et al., “Implantable micromechanical parylene-based pressure sensors for unpowered intraocular pressure sensing,” J. Micromechanics Microengineering, vol. 17, no. 10, pp. 1931–1938, Aug. 2007, doi: 10.1088/0960-1317/17/10/002.
[4] R. Caldwell, M. G. Street, R. Sharma, P. Takmakov, B. Baker, and L. Rieth, “Characterization of Parylene-C degradation mechanisms: In vitro reactive accelerated aging model compared to multiyear in vivo implantation,” Biomaterials, vol. 232, p. 119731, Feb. 2020, doi: 10.1016/j.biomaterials.2019.119731.
[5] C.-C. Wu et al., “A pliable and batteryless real-time ECG monitoring system-in-a-patch,” in VLSI Design, Automation and Test(VLSI-DAT), Apr. 2015, pp. 1–4, doi: 10.1109/VLSI-DAT.2015.7114521.
[6] M. Cieślik et al., “Engineering of bone fixation metal implants biointerface—Application of parylene C as versatile protective coating,” Mater. Sci. Eng. C, vol. 32, no. 8, pp. 2431–2435, Dec. 2012, doi: 10.1016/j.msec.2012.07.018.
[7] M. Cieślik, M. Kot, W. Reczyński, K. Engvall, W. Rakowski, and A. Kotarba, “Parylene coatings on stainless steel 316L surface for medical applications — Mechanical and protective properties,” Mater. Sci. Eng. C, vol. 32, no. 1, pp. 31–35, Jan. 2012, doi: 10.1016/j.msec.2011.09.007.
[8] M. Kazemi et al., “A test microchip for evaluation of hermetic packaging technology for biomedical prosthetic implants,” in The 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 2004, vol. 4, pp. 4093–4095, doi: 10.1109/IEMBS.2004.1404142.
[9] A. Lecomte, A. Degache, E. Descamps, L. Dahan, and C. Bergaud, “In vitro and in vivo biostability assessment of chronically-implanted Parylene C neural sensors,” Sens. Actuators B Chem., vol. 251, pp. 1001–1008, Nov. 2017, doi: 10.1016/j.snb.2017.05.057.
[10] W. R. Dolbier and W. F. Beach, “Parylene-AF4: a polymer with exceptional dielectric and thermal properties,” J. Fluor. Chem., vol. 122, no. 1, pp. 97–104, Jul. 2003, doi: 10.1016/S0022-1139(03)00100-3.
Comments
Homepage 4/17/2020. 10:17:10 AM
... [Trackback] [...] Informations on that Topic: blog.paryleneconformalcoating.com/whats-the-difference-between-potting-and-conformal-coating/ [...]

londondrugscanada.bigcartel.comlondon-drugs 4/17/2020. 10:17:10 AM
cialis uk https://londondrugscanada.bigcartel.com/london-drugs This is nicely expressed. !