Parylene Protects Stents
Posted by Sean Horn
Friday, December 16, 2016 7:48
@ 7:48 AM
Biocompatible parylene conformal coatings provide superior protection for medical stents. They represent an enabling technology consistently applied to medical devices of all types for 35 years, to diminish problems stemming from surface microporosity and consequent biofluid corrosion after implant. Providing a reliable barrier to chemicals and moisture, parylene’s static and dynamic coefficients of friction are comparable to those of Teflon.
Implanted parylene devices have demonstrated:
- effective nonthrombogenic properties,
- excellent hemocompatibility,
- exceptional resistance to potentially damaging effects of bodily fluids, electrolytes, enzymes, lipids, and proteins,
- good dry-film lubricity,
- limited toxicological responses, and
- minimal incidence of fibrous capsule formation,
- without producing immunological responses.
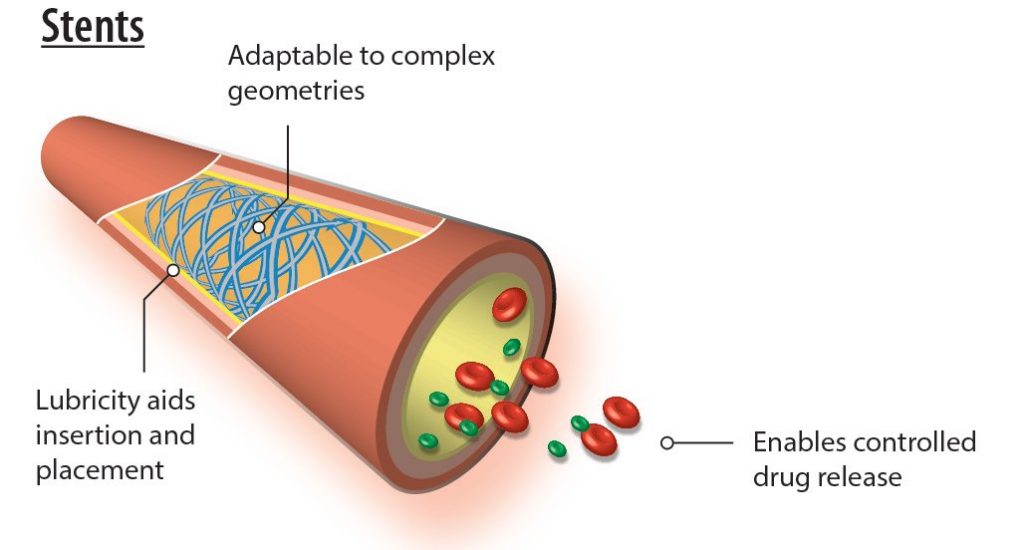
Applicability of Parylene for Stents
Chemically inert, parylene provides a superior conformal coating for stents, among the most valuable of implantable medical devices. Its use meets all professional requirements, including the USP Class VI implantable plastic material standard, conforming to both the ISO 10993 biocompatibility standard and RoHS standards.
Medically-implanted stents require a finely-conceived and produced geometrical configuration to be successful. Parylene adapts to these product requirements; its appropriate conformability characteristics result from its unique chemical vapor deposition (CVD) application process. Enacted in a specialized vacuum chamber, CVD:
- enables parylene to coat inside seemingly closed structures,
- including the coating’s penetration into surface openings at the micron/individual molecule level, while
- completely and effectively encapsulating the stent’s external features with a resilient protective film.
CVD processing also eliminates formation of undesirable air pockets along and within the coating’s surface, a major drawback of liquid coatings such as acrylic, epoxy, silicone and urethane. With parylene, there is neither bridging nor over-filming of minute surface openings, a drawback commonly occurring with wet coating materials. Since parylene generates a gaseous, molecule-by-molecule growth that adheres to the stent’s every contour, the truly conformal coating that results penetrates the stent’s surface microcrevices, and prevents corrosion from harsh bodily fluids after deployment. In addition, the galvanic electrochemical reaction that can develop when dissimilar metals like nitinol and platinum are combined within a single stent are largely eliminated by parylene’s excellent moisture barrier properties, improving the stent’s function while prolonging its operating life.
In addition, parylene’s micron-thin coatings offer this exceptional resilience and protection to implanted stents without adding significant dimension or weight. This feature is an added benefit for internal body medicine and can be easily adapted for MEMS/nano purposes. Present research and development focused on controlling coating dimension and weight should lead to the creation of stents that are simultaneously smaller and more complex, with enhanced capacity for medical treatment. Only CVD-applied parylene generates these specialized service features.
Among other advantageous properties for use with stents, parylene conformal films:
- are composed without catalysts, fillers, plasticizers, solvents or stabilizers,
- require no additional curing, cross-linking or related processes after CVD is complete,
- are pinhole-free with excellent protective barrier properties,
- create micro-thin, transparent films of precisely controlled thickness, free of imperfections,
- are hydrophobic,
- have no toxic outgassing,
- can deliver drugs from the stent to the implanted body region,
- are chemically inert and non-reactant with the carrier polymers or drugs,
- have no leachable or components, and
- can be used as a pre-coat primer for other stent coating materials if necessary.
Parylene conformal films can be precisely deposited on virtually all available stent materials, simultaneously serving as both a carrier for drug delivery and a mechanism for controlled pharmaceutical release.
For widely-used drug-eluting stents, parylene films significantly diminish the incidence of restenosis, the re-narrowing of arteries, for most patients. Typically, treatment drugs are applied over a parylene-coated stent substrate. This pharmaceutical layer is then treated with another coating of micro-thin, partially porous parylene, which permits its introduction into the patient’s system at a regulated level. In some cases, drugs and parylene are deployed in multiple, alternative layers for longer-term treatment effectiveness.
Future Improvements Inevitable
Parylene is well-suited for use with in vivo devices such as stents. Coating virtually any surface that is vacuum-stable conformally and uniformly through CVD, parylene stands out among conformal materials as a superior coating for stents’ often intricate structures, achieved without pooling or webbing. Its versatility continually leads to new applications:
- As a bonding, or tie-connection layer, parylene coatings are now applied between the metal stent and the drug/drug-carrier polymer.
- As a drug-release control agent, parylene is applied over the drug-coated stent.
- Development of newer biocompatible variants of parylene should stimulate additional medical applications, for stents and other implantable devices, while lowering production costs.
This combination of medical benefits and healthcare cost savings coupled with a lower per-unit cost will likely stimulate further development of better quality stents, protected by parylene conformal coatings.
To learn more about medical devices and parylene, download our whitepaper:
The Truth about Medical Devices and Parylene
Comments
Homepage 4/17/2020. 10:17:10 AM
... [Trackback] [...] Informations on that Topic: blog.paryleneconformalcoating.com/whats-the-difference-between-potting-and-conformal-coating/ [...]

londondrugscanada.bigcartel.comlondon-drugs 4/17/2020. 10:17:10 AM
cialis uk https://londondrugscanada.bigcartel.com/london-drugs This is nicely expressed. !