Types of Parylene
Posted by Sean Horn
Friday, March 10, 2017 7:34
@ 7:34 AM
Applied in a gaseous form to component surfaces through a chemical vapor deposition (CVD) process, parylene (Poly-para-xylylene) films protect printed circuit boards (PCBs) and similar electrical assemblies. Gaseous CVD application supports efficient coating of complex component surfaces characterized by crevices, exposed internal areas, or sharp edges. Depending on the specific use, parylene conformal coatings can be effective in the range of 0.1 – 76 microns’ thickness, far finer than competing coating materials. Equally as strong, adaptable and versatile parylene protects substrates with
- pinhole-free coverage at thickness levels finer than other materials,
- a completely homogeneous surface, with thermal stability between -200 °C to +125°C,
- exceptional electrical insulation,
- low dielectric constancy and
- high tension strain
These properties assure the deposited films reliably resist the incursion of acids, bodily fluids, caustic solutions, dust, temperature extremes, water vapor, and numerous other contaminants. In addition to consumer and industrial products, conformal coatings have a wide range of aerospace, automotive, military, and medically biocompatible/implantable device uses. Prominent parylene types include varieties C, N, F, D, and AF-4; each has a range of distinctive uses distinguished by their relatively unique chemical, electrical and physical properties.
Parylene Types and Their Uses
Each specific parylene type is valuable for properties unique to the type; among the most prominent are:
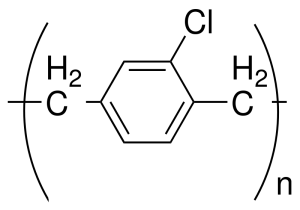 Parylene C: Type C is used as a conformal coating more frequently than any parylene variety. A poly-monochoro para-xylene, produced from dimer material, C can be deposited quickly at room temperature on most substrates via CVD. Its main-chain phenyl ring has one chlorine group per repeat-unit, making it a carbon-hydrogen combination material. With low chemical, moisture, and vapor permeability, Type C films are characterized by outstanding defense against corrosive gases, and ongoing assembly protection in oxygen-dominated atmospheres for 100,000 hours (10 years), at temperatures of 100° C (212° F), water’s boiling point. Moreover, at 49.4° C/10 -6 torr (1 torr = 1/760 of the standard atmospheric pressure [SAP = 1 mm Hg]), C demonstrates performance ratings of 0.12% total weight-loss, confirming its reliable vacuum stability as a conformal coating.
Parylene C: Type C is used as a conformal coating more frequently than any parylene variety. A poly-monochoro para-xylene, produced from dimer material, C can be deposited quickly at room temperature on most substrates via CVD. Its main-chain phenyl ring has one chlorine group per repeat-unit, making it a carbon-hydrogen combination material. With low chemical, moisture, and vapor permeability, Type C films are characterized by outstanding defense against corrosive gases, and ongoing assembly protection in oxygen-dominated atmospheres for 100,000 hours (10 years), at temperatures of 100° C (212° F), water’s boiling point. Moreover, at 49.4° C/10 -6 torr (1 torr = 1/760 of the standard atmospheric pressure [SAP = 1 mm Hg]), C demonstrates performance ratings of 0.12% total weight-loss, confirming its reliable vacuum stability as a conformal coating.- Parylene N: Less frequently used than Parylene C, Type N assumes a completely linear format, making it the most natural member of the parylene series. Highly crystalline, each Type N molecule consists of a carbon-hydrogen combination. Its melting point of 420° C exceeds other parylene types; exceptional vacuum stability is evidenced by total weight-loss levels of 0.30% at 49.4° C, and 10 -6 torr. Higher frequency applications are encouraged because of N’s low dielectric constant and dissipation values, in comparison to other parylenes.
Types C and N exhibit faster deposition rates than most other parylenes, making them useful for a wider range of uses. They also display generally lower levels of trace metal contamination and exceptional resistance to the destructive impact of corrosive chemicals. A third parylene, Type F, has fluorine atoms on its aromatic ring, distinguishing it from Types C and N. Other salient characteristics include:
- Parylene F: Type F is fluorinated, with markedly diminished coating capacitation compared to other parylene types. The consequent lower electrical charge during operation generates a reduced dielectric constant and better thermal stability, enhancing its functionality for inner layer dielectric (ILD) applications; these properties expand its utility for ultra large scale integration (ULSI), wherein one million or more circuit elements are situated on a single chip, supporting both microelectromechanical systems (MEMS) and nanotech (NT) applications. F exhibits high coating density, more throw-capability and substantially greater penetrating power than other parylenes.
Other prominent parylene types are:
- Parylene D: Originating from the same monomer as Type C, Parylene D’s chemical composition contains two atoms of chlorine in place of two hydrogen atoms. Nevertheless, unlike C, D lacks sufficient biocompatibility for wide use in medical devices. Withstanding temperatures in excess of 125° C, D offers effective coating protection at 134° C (273° F), providing reliable assembly protection for 10 years at 100° C, in oxygen-dominated environs.
- Parylene AF-4: With a precursor requiring a three-step synthesis process, AF-4 is more costly to manufacture; overall yields are limited compared to other parylenes. A Teflon (PTFE) analogue, its higher oxidative resistance and greater UV stability allow for specialized applications.
Conclusion
Vacuum CVD causes parylene to condense on substrate surfaces in a polycrystalline fashion, generating a truly conformal film that is pinhole free and long-lasting. Since other parylene types have deposition rates that are slower and more expensive, most current parylene applications use Types N and C. Although Type N is chemically the most fundamental of the parylenes, Type C is more widely applied as a product/component coating.
To learn more about the basics of parylene, download our whitepaper:
{{cta(‘a28f6b94-0c55-4b10-9b98-95ab73c76740’)}}
Comments
Homepage 4/17/2020. 10:17:10 AM
... [Trackback] [...] Informations on that Topic: blog.paryleneconformalcoating.com/whats-the-difference-between-potting-and-conformal-coating/ [...]

londondrugscanada.bigcartel.comlondon-drugs 4/17/2020. 10:17:10 AM
cialis uk https://londondrugscanada.bigcartel.com/london-drugs This is nicely expressed. !