Understanding Parylene Deposition
Posted by Sean Horn
Friday, July 31, 2015 8:10
@ 8:10 AM
Parylene’s deposition process is unique among conformal coatings. Unlike others that start as a liquid, get deposited and dry, it starts as a solid. Parylene coating equipment turns it into a vapor, where it then deposits onto the substrate. This unique four-step method poses some challenges but also brings real advantages.
Sublimation
In the first step of the process, the powdery raw material, called dimer, is heated. As the temperature approaches 150 degrees Celsius, the dimer sublimates into a gas without passing through a liquid state. At this point, the dimeric gas is ready to pass into the next stage of the deposition process.
Pyrolysis
The dimer gas actually consists of two parylene molecules, bonded together. In the pyrolysis stage, the gas is further heated in a furnace to temperatures as high as 700 degrees Celsius. The heat splits the molecules apart — creating a monomer. That monomeric vapor is what becomes the parylene conformal coating when it reaches the item to be coated in the deposition stage.
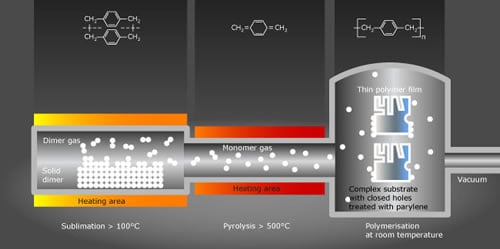
Deposition and Polymerization
The container holding the items to be coated is depressurized to create a vacuum. That vacuum sucks the parylene gas out of the pyrolysis chamber and into the deposition chamber. In that chamber, which is maintained at normal room temperature, the parylene vapor turns into a polymer, coating everything that it touches. During the deposition process, the parylene gas cools without having any meaningful heating effect beyond a few degrees on the items being coated.
Waste Collection
The vacuum pump that pulls the parylene gas through the deposition chamber also plays an important role in disposing of waste vapor. As the gas exits the chamber, the pump pulls it though a cold trap. That trap, which is usually cooled to extreme subzero temperatures, causes the parylene to solidify so that it can be disposed of.
Challenges in the Process
Adhesion is generally one of the largest problems with parylene, especially when trying to coat noble metals. This is often addressed by applying silane A-174 or other adhesion promoters prior to or during the deposition process.
In addition, because of parylene’s unique method of deposition, quantities can be limited. Put simply, you can only coat as many items as will fit in the deposition system’s coating chamber. While these systems can be relatively large, there are still practical limits to how many items you can coat at one time.
Furthermore, while parylene coatings are typically thin, they also deposit relatively slowly. The fastest-depositing variant of parylene — Parylene C — typically deposits at a rate of 0.2 mils or 5 microns per hour. This means that a 75 micron coating would take approximately 15 hours. Parylene N and D deposit more slowly.
The Power of Vapor
Even given some of the drawbacks inherent in the parylene deposition process, using a vapor-phase coating has significant advantages. For example, because the vapor is essentially dry and because the process occurs at room temperature, even very sensitive items can be safely coated without any damage.
Parylene vapor can go anywhere that air can. This means that it coats under parts, inside small crevices and can even coat inside semi-sealed areas. Its thinness means that it can coat without forming bridges in tight areas as well.
A parylene coating is also truly conformal, since it does not run or drip on the surface the way that some liquid coatings can. Since it deposits on the entire surface at the same time, it also creates an extremely even coating of the same thickness, everywhere. This characteristic is one of the reasons that it is also typically the most pinhole free of all of the conformal coating compounds.
Ultimately, the parylene deposition process is an important part of what makes parylene such a desirable coating choice. While its vapor-phase deposition can be time-consuming and expensive relative to other coatings and their more traditional application methods, its results are also unparalleled for many applications.
Diamond-MT provides high-quality parylene conformal coating services for all industries, including military, electronics, and medical applications. Contact us today at 1-814-535-3505 to discuss your next project with our team of experts.
Download our guide on Parylene 101
Comments
Homepage 4/17/2020. 10:17:10 AM
... [Trackback] [...] Informations on that Topic: blog.paryleneconformalcoating.com/whats-the-difference-between-potting-and-conformal-coating/ [...]

londondrugscanada.bigcartel.comlondon-drugs 4/17/2020. 10:17:10 AM
cialis uk https://londondrugscanada.bigcartel.com/london-drugs This is nicely expressed. !